Originality is not easily achieved. If you wish to republish this article, please contact the staff and provide proper attribution to the source in the republished content. Otherwise, infringement will be subject to legal action.
In catalytic reactions, certain chemical transformations may involve one or more reaction steps, where the products of the first step can serve as reactants for the second step. This is commonly referred to as a cascade catalytic reaction.
A cascade catalytic reaction refers to the process in which two or more independent reactions occur successively within a catalytic reaction system.
During a cascade catalytic reaction, there is no need for additional separation and purification steps. This can reduce the number of reaction steps, energy consumption, and the production of by-products, leading to maximum savings in reactant usage and effective enhancement of the efficiency of catalytic reactions.
In cascade catalytic reactions, all catalysts are introduced into the catalytic reaction system during the experimental preparation stage. There is no need to add catalysts during the catalytic reaction process, effectively optimizing complex reaction processes.
In a cascade catalytic reaction, the term "cascade" can have two meanings:
① Simultaneous action of two different catalytic reaction mechanisms;
② Catalytic reactions taking place one after another in a consecutive manner.
Cascade catalytic reactions are typically divided into:
Orthogonal Cascade Catalytic Reactions
Self-Cascade Catalytic Reactions
Assisted Cascade Catalytic Reactions
1. Orthogonal Cascade Catalytic Reactions
The distinguishing feature of orthogonal cascade catalytic reactions is their mutual independence. In orthogonal catalytic processes, two or more different catalytic agents are involved, each with distinct functions that do not interfere with one another. As depicted in Figure 1, for instance, reactant A reacts with catalyst A to produce product A. Subsequently, in the next stage of reaction, product A acts as reactant B and engages with catalyst B to yield product B.
In orthogonal cascade catalytic reactions, once catalytic reaction A occurs and generates product A, both catalytic reactions proceed simultaneously. The inclusion of two or more catalytic agents in the entire reaction system of orthogonal cascade catalysis effectively prevents limitations that may arise from the use of a single catalyst.
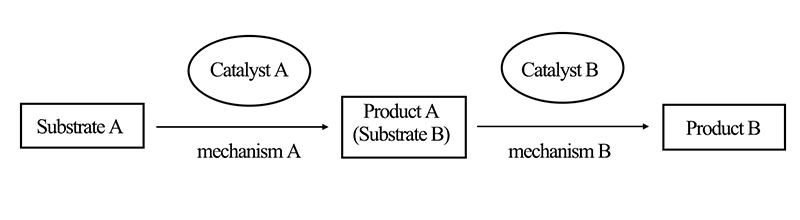
Figure 1. Schematic Diagram of Orthogonal Cascade Catalytic Reactions
2. Self-Cascade Catalytic Reactions
Self-cascade catalytic reactions refer to reactions conducted using a single catalyst, where the reaction process involves two or more different reaction mechanisms. In self-cascade catalytic reactions, all necessary reactants and catalysts are preloaded into the reaction system before initiating the process. Once the first catalytic reaction is triggered, subsequent reactions occur spontaneously in succession. As illustrated in Figure 2, for example, reactant A is transformed into product A through the action of catalyst A. Subsequently, product A acts as reactant B in the next reaction and is further transformed into product B by catalyst A.
In an ideal scenario, self-cascade catalytic reactions proceed sequentially based on the transformation of reactant molecules. However, on a macroscopic level, these reactions occur simultaneously.
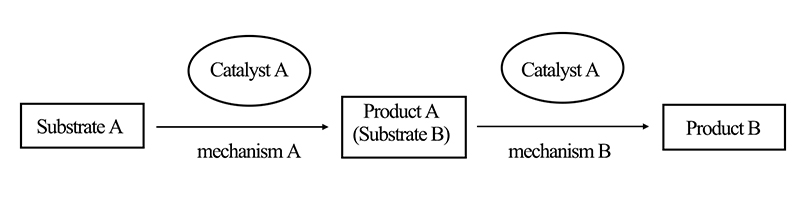
Figure 2. Schematic Diagram of Self-Cascade Catalytic Reactions
3. Assisted Cascade Catalytic Reactions
Assisted cascade catalytic reactions involve modifying the original reaction conditions during the reaction process to alter the catalytic reaction mechanism. This approach aims to expand the catalytic conversion range, performance, and selectivity of a single catalyst. As depicted in Figure 3, catalyst A can complete the first step of the catalytic reaction process. Upon introducing an auxiliary reagent into the reaction system, catalyst A transforms into catalyst B, which then catalyzes the conversion of reactants.
Unlike self-cascade catalytic reactions, in assisted cascade catalytic reactions, the two reactions cannot occur simultaneously.
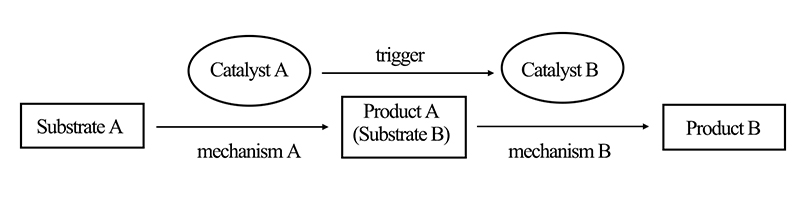
Figure 3. Schematic Diagram of Assisted Cascade Catalytic Reactions
For various types of cascade catalytic reactions mentioned above, Perfectlight has introduced the PLS-MRRS Catalytic Reaction System designed specifically for heterogeneous cascade catalytic reactions. This system is suitable for catalytic reaction types such as methane reforming, methanol-water reforming, CO2 reduction, hydrogenation reactions, Fischer-Tropsch synthesis, VOC degradation, and CO oxidation, among others. The PLS-MRRS Catalytic Reaction System employs a fixed-bed reactor configuration, allowing both gas and liquid to participate in reactions simultaneously. Depending on the specific reaction requirements, users can choose a single reactor or multiple reactors. Currently, the system can be expanded to accommodate up to four levels of cascade reactors.

Note: The explanation of cascade catalytic reactions is based on the translation and compilation of referenced articles. If any mistakes are present, corrections are welcome.
[1] Deryn E. Fogg∗, Eduardo N. dos Santos∗, Tandem catalysis: a taxonomy and illustrative review[J]. Coordination Chemistry Reviews, 2004, 248: 2365.
[2] Niladri Maity, Srikanta Patra∗, Recent advances in (hetero)dimetallic systems towards tandem catalysis[J]. Coordination Chemistry Reviews, 2021, 434: 213803.
[3] Yahya Gambo, Sagir Adamu, Mohammad M. Hossain∗ et al., Tandem catalysis: A sustainable alternative for direct hydrogenation of CO2 to light olefins[J]. Applied Catalysis A, General, 2022, 641: 118658
Recommended
Solution