Chromatography is a separation and analytical technique that utilizes the different distribution coefficients of substances in the mobile phase and the stationary phase. When the mobile phase and the stationary phase undergo relative motion, different substances in the mobile phase can be separated within the stationary phase, as shown in Figure 1.
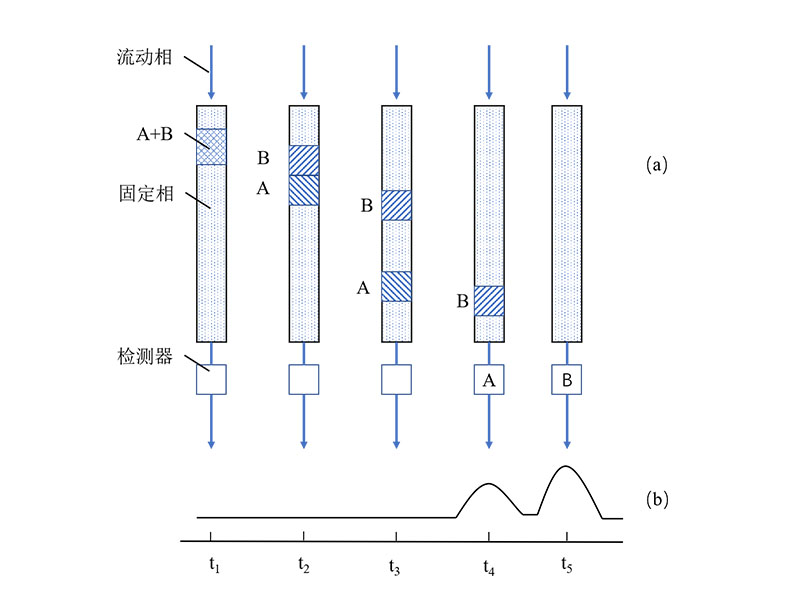
Figure 1 Separation of Binary Mixture[1]
(a) Elution process inside the column, (b) Chromatogram recorded
The chromatography column is the heart of the chromatograph, installed inside a temperature-controlled column chamber. There are two main types of chromatography columns: packed columns and open tubular columns (also known as capillary columns).
Packed columns are made of materials like stainless steel or glass and are filled with an appropriate stationary phase based on analytical requirements. They typically have an outer diameter of 3 to 4 mm and a length of up to 5 meters.
Capillary columns, made of quartz, have their stationary phase coated on the inner walls of the capillary or bonded to the tube walls through certain chemical processes. Their inner diameter generally does not exceed 0.53 mm, and the length can range from tens of meters to hundreds of meters.
The chromatography column's nameplate displays relevant parameters such as outer diameter, length, maximum temperature resistance, etc. When replacing a chromatography column, you can refer to the original specifications and model number.
The selection of the outer diameter and length of the chromatography column depends on the desired separation effect. This is especially crucial in chromatographic analysis methods involving multiple columns and valves.
The selection of chromatography column type is based on the substances to be separated:
① For separating hydrogen, oxygen, and nitrogen gases, 5A molecular sieve can be chosen;
② For separating carbon monoxide and carbon dioxide, TDX-01 packed column can be chosen;
③ For separating small molecules such as alcohols, aldehydes, and acids, INNOWAX capillary column can be chosen.


Figure 2 Capillary Column (left) and Packed Column (right)