Original content is not easy; if you wish to reproduce this article, please contact the staff and provide proper attribution in the republished article; otherwise, it will be treated as copyright infringement!
Traditional organic synthesis steps are often cumbersome due to thermodynamic constraints, requiring a substantial amount of energy and organic solvents in the reaction process. Additionally, the selectivity of products is often low, and the generated byproducts can be environmentally harmful. Some organic reactions with unique structures in the total synthesis process can be significantly simplified and made more environmentally friendly by using photocatalytic reactions.
In recent years, with the rapid development of photocatalysis in organic synthesis, photocatalytic organic synthesis has gradually become an important branch of the field of photocatalysis.
Currently, related research primarily involves several important organic reactions, including coupling reactions, polymerization reactions, oxidation reactions of functional groups, reduction reactions of functional groups, and the synthesis of chiral molecules.
As research progresses, photocatalysts are no longer limited to traditional semiconductor photocatalysts. Common alternative photocatalysts include ruthenium, iridium metal-organic complexes, dyes, and more.
The increasing attention to photocatalytic organic synthesis is primarily due to the following advantages when applying photocatalytic technology to organic synthesis reactions:
Using inexhaustible solar energy as an energy source can save traditional energy waste and associated pollution;
The reaction conditions are mild and can be conducted at room temperature and pressure, reducing the use of toxic and corrosive reagents, and lower temperatures are more favorable for the synthesis of chiral compounds;
By designing photocatalysts, the selectivity of the target product can be effectively improved, reducing the generation of byproducts and enhancing atomic economy;
The free radical reaction mechanism of photocatalytic organic synthesis can overcome the limitations of thermodynamic reactions in traditional organic synthesis, simplifying multi-step reactions into one-step reactions, significantly reducing energy, solvent, reagent consumption, and synthetic waste production.
There are many important factors influencing photochemical synthesis reactions, with the main ones listed below:
1. Catalyst selection and wavelength of light
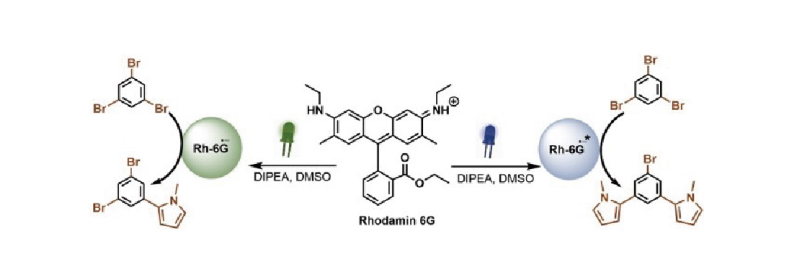
Photocatalysts absorb photons of different wavelengths, leading to different excited states and redox energies, which may result in differences in chemical reactions. Selecting the appropriate wavelength of light can effectively reduce side reactions and improve product selectivity.
Perfectlight LED light sources offer a choice of multiple wavelengths ranging from 255 to 940 nm.
Blue light absorption regions λ/nm: 450, 460, 475, 485, 505
Green light absorption regions λ/nm: 520, 525, 535, 550, 575
2. Light intensity and irradiation efficiency
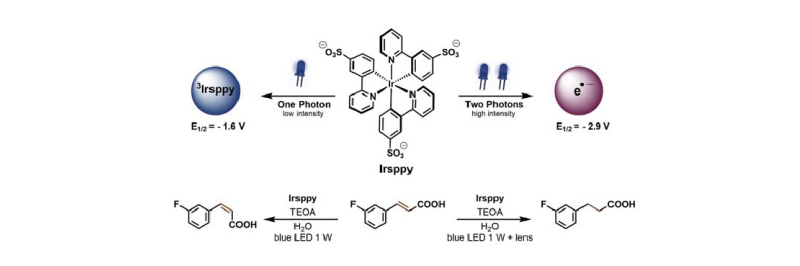
When the light intensity is much higher than the concentration of photocatalysts, the concentration of excited photocatalysts in the excited state increases. Excited photocatalysts can absorb a second photon, leading to a higher-energy excited state or high-energy electron overflow, initiating new reactions. Adjusting the appropriate light intensity can control the type of reaction.
Perfectlight LED light sources offer adjustable light power ranging from 100 to 450 mW/cm2.
3. Reaction apparatus
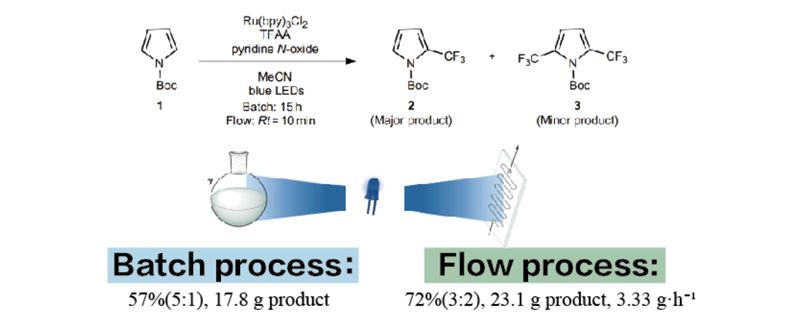
Traditional kettle-type reactions are convenient but limited in terms of light penetration efficiency, which can limit reaction rates. Additionally, low mass transfer and heat transfer efficiencies result in long reaction times and low selectivity. New types of flow chemistry reactions can significantly enhance light efficiency, accelerate mass transfer and heat transfer rates, improving yield and selectivity. Flow reactions are also easier to scale up for production.
Perfectlight PLR-SMCR1000 Multiphase Microchannel Reaction System can be used for small-scale photocatalytic flow experiments.
4. Residence time
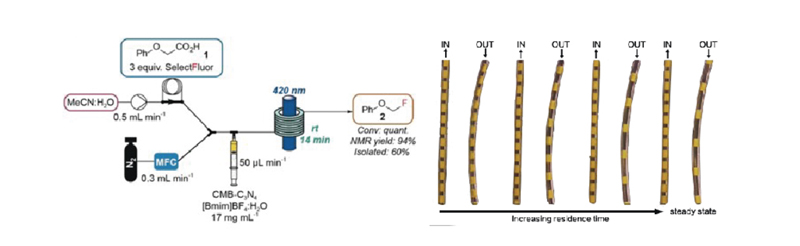
Residence time, in the context of flow chemistry, refers to the time the reaction mixture is exposed to light. Too short a residence time may result in incomplete reactions, while too long a residence time may lead to side reactions. The right residence time can improve conversion rates and reduce side reactions.
Perfectlight PLR-SMCR1000 Multiphase Microchannel Reaction System offers reaction tubes with volumes of 5 mL and 10 mL, as well as flow rates ranging from 1.5 to 8.5 mL/min to meet various residence time requirements.
5. Flow rate and mixing efficiency of the reaction solution
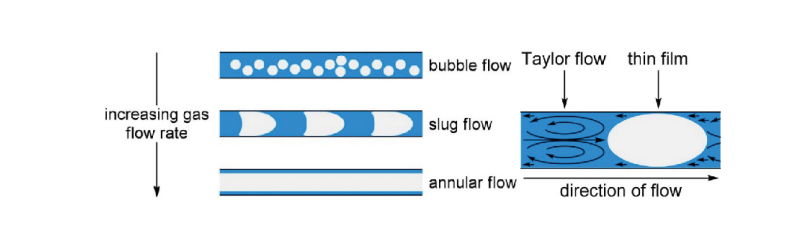
Flow reactions can rapidly mix reactants and catalysts, improving mixing efficiency. Gas-liquid and liquid-liquid Taylor fluids can further promote in-phase mixing rates, increase the contact area between two phases, and accelerate the reaction rate.
The Perfectlight multiphase microchannel reaction system can introduce gas-liquid and liquid-liquid Taylor fluids to further enhance in-phase mixing rates, especially suitable for gas/liquid and liquid/liquid heterogeneous reaction systems.
The above content is translated and summarized by the author based on reference literature. The author's proficiency is limited, so please feel free to correct any errors!